Covalent Bonding
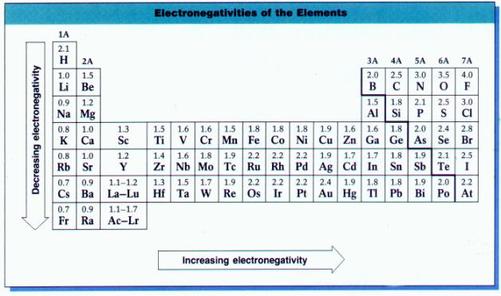
Electronegativity is a measure of the attraction of an atom for electrons in a covalent bond.
The difference in electronegativities of two elements can be used to predict the nature of the chemical bond. Bond type can be described as belonging to one of three classes:
Exceptions: HF, BF3, and BF2. These are polar covalent.
Polar covalent bonds indicate that a dipole exists. A dipole is a molecule with a partially positive (d+) and a partially negative (d-) charge. Atoms with greater electronegativity will have a partially negative charge.
The difference in electronegativities of two elements can be used to predict the nature of the chemical bond. Bond type can be described as belonging to one of three classes:
- nonpolar covalent
- polar covalent
- ionic
Exceptions: HF, BF3, and BF2. These are polar covalent.
Polar covalent bonds indicate that a dipole exists. A dipole is a molecule with a partially positive (d+) and a partially negative (d-) charge. Atoms with greater electronegativity will have a partially negative charge.